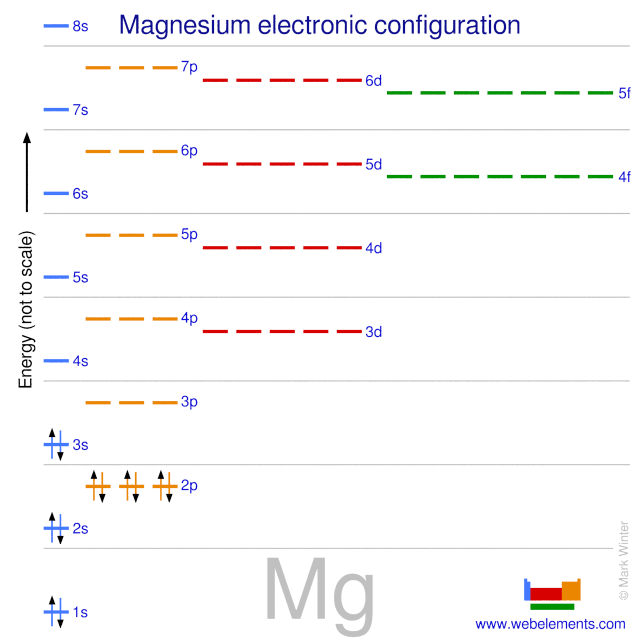
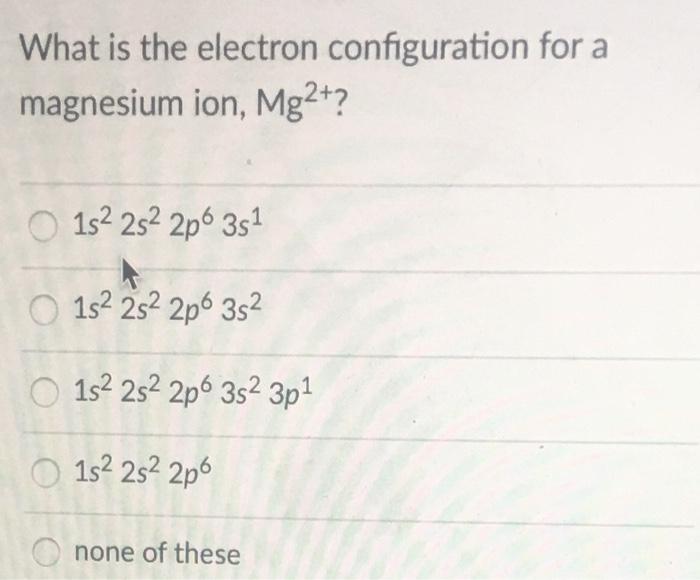Starting with the atomic electron configurations of Mg and N to forming the corresponding | Homework.Study.com

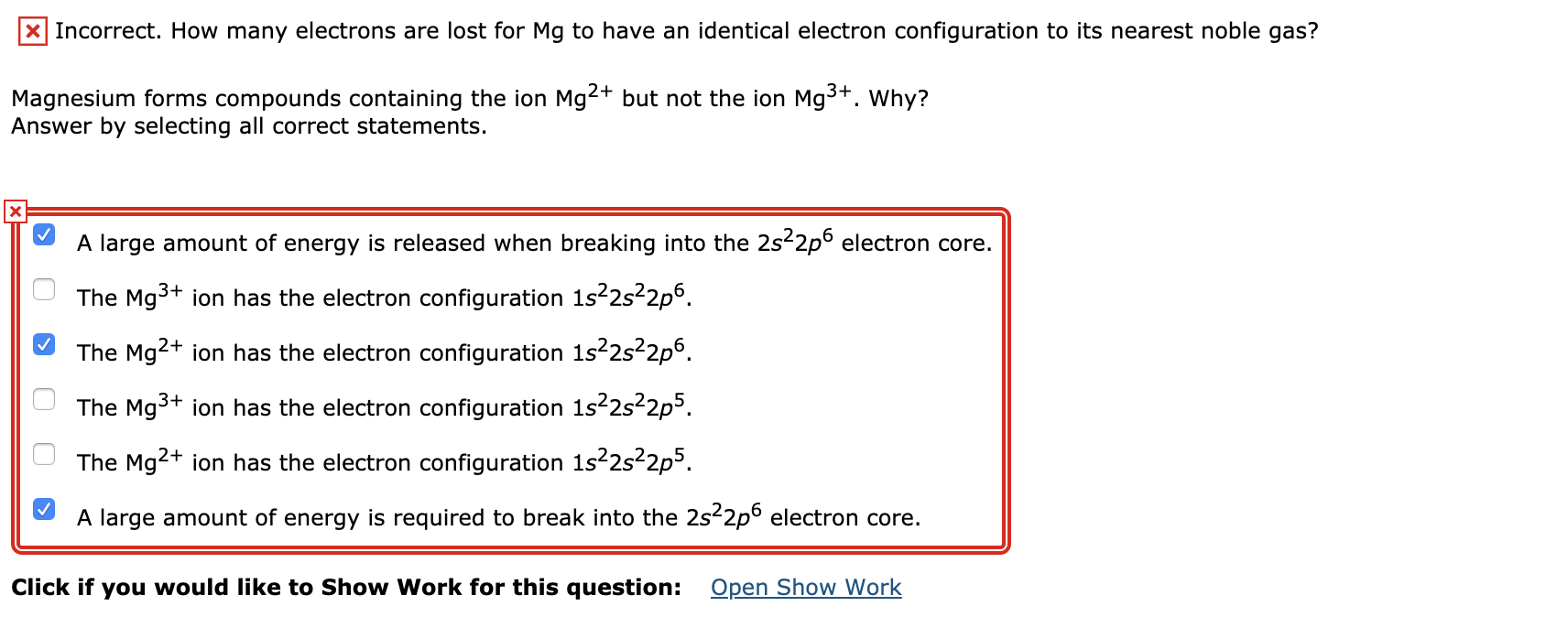
SOLVED: The electron configuration of Mg2+ ion is A 1s2 252 2p6 B. 152 252 2p6 352 3p2 C 1s2 252 2p4 D. 1s2 2s2 2p6 352

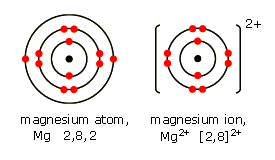
a). Write down the electronic configuration of (i) magnesium atom, and (ii) magnesium ion, (At No. of Mg=12) ,brgt (b). Write down the electronic configuration of (i) sulphur atom, and (ii) sulphide
Hat is the charge of the ion most likely to be formed from Magn to achieve noble gas notation? | Socratic